The U.S. Food and Drug Administration (FDA) has released information for entry line refusals for the month of March. The agency reported refusing a total of 208 seafood entry lines last month. Of these, 17 (8.2%) were of shrimp entry lines refused for reasons related to banned antibiotics.
The number of shrimp entry lines refused for banned antibiotics last month was more than the double the total amount of shrimp entry lines refused for this reason in January and February combined.
The seventeen entry line refusals in March involved seven companies from three different countries and were reported by six different regional offices of the FDA:
- RDR Exports (India), a company not currently listed on Import Alert 16-124 for chloramphenicol, but listed on Import Alert 16-129 for nitrofurans on October 9, 2015, had four entry lines refused for shrimp contaminated with nitrofurans in the Florida District;
- Five Star Marine Exports Pvt. Limited (India), a company not currently listed on Import Alert 16-124 for chloramphenicol, but listed twice, at two different addresses, on Import Alert 16-129 for nitrofurans on October 9, 2015, had three entry lines refused for shrimp contaminated with veterinary drug residues and nitrofurans in the New York District;
- Kay Kay Exports (India), a company not currently listed on Import Alert 16-124 for chloramphenicol, but listed on Import Alert 16-129 for nitrofurans on September 17, 2015, had two entry lines refused for shrimp contaminated with veterinary drug residues and nitrofurans in the Los Angeles District;
- Minh Phu Hau Giang Seafood Corp. (Vietnam), a company not currently listed on either Import Alert 16-124 or Import Alert 16-129, had one entry line refused for shrimp contaminated with veterinary drug residues in the Southwest District;
- Bac Lieu Fisheries (Vietnam), a company not currently listed on either Import Alert 16-129, but listed on Import Alert 16-124 for sulfadimethoxine and sulfadiazine on June 19, 2015, had one entry line refused for shrimp contaminated with veterinary drug residues in the Atlanta District;
- Zhou Shan Xin Ming Zhou Aquatic Products Co. Ltd. (China), a company that has not been exempted from Import Alert 16-131, had one entry line refused for shrimp contaminated with veterinary drug residues in the New York District;
- Zhanjiang Regal Integrated Marine Resources Co., Ltd. (China), a company that had been exempted from Import Alert 16-131 in June 2015, had four entry lines refused for shrimp contaminated with veterinary drug residues in the Southwest District and one entry line refused for shrimp contaminated with nitrofurans in the Los Angeles District. Zhanjiang Regal‘s exemption from Import Alert 16-131 was removed on February 29, 2016 and its shipments of shrimp to the United States are currently subject to detention without physical examination.
Prior to losing its exemption from Import Alert 16-131 at the end of February, Zhanjiang Regal had significantly increased its shipments of shrimp to the U.S. market. Those shipments were largely made to corporate entities affiliated with companies that had previously imported large quantities of Malaysian shrimp before those shipments collapsed last year.
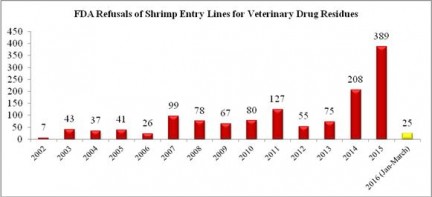
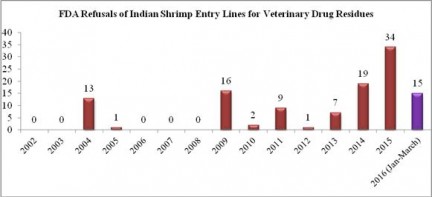
The majority of shrimp entry line refusals in March were for shipments from India. Last year, a record total of 34 entry lines of shrimp from India were refused for reasons related to banned antibiotics. This amount was exactly half the total amount (68) of entry lines of shrimp from India refused over the previous thirteen years combined. In the first three months of 2016, the FDA has already reported refusing 15 entry lines of Indian shrimp. As shown in the chart of historical refusals of Indian shrimp entry lines, this means that through March of this year, the FDA has already refused more Indian shrimp contaminated with antibiotics than in all but three (2009, 2014, and 2015) of the last fourteen years.