News Alert: Shipments of Vietnamese Shrimp Contaminated with Antibiotics Discovered by FDA Again in November
Today, the U.S. Food and Drug Administration (FDA) published information regarding entry line refusals for the month of November. The agency reported refusing a total of 129 seafood entry lines last month. Of these, 9 (7.0%) were of shrimp entry lines refused for reasons related to banned antibiotics.
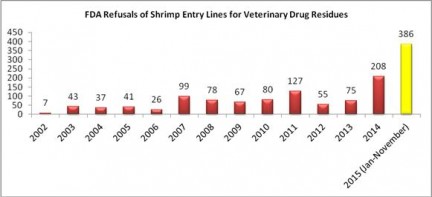
With one month left in 2015, the FDA has now refused a record total of 386 entry lines of shrimp products for reasons related to banned antibiotics.
The nine entry line refusals in November principally involved shrimp manufactured by one Vietnamese company. The refusals were reported from four different FDA District Offices spread across the country:
- Bac Lieu Fisheries (Vietnam), a company listed on Import Alert 16-124 for sulfadoxine on June 19, 2015 but not currently listed on Import Alert 16-129, had five entry lines refused for shrimp contaminated with veterinary drug residues in the Los Angeles District, two entry lines refused for shrimp contaminated with veterinary drug residues in that Atlanta District, and one entry line refused for shrimp contaminated with veterinary drug residues in the New York District; and
- Globe Season Enterprises (Malaysia), a company not currently listed on either Import Alert 16-124 or Import Alert 16-129, had one entry line refused for shrimp contaminated with veterinary drug residues in the Baltimore District.
The eight entry line refusals of shrimp shipped from Vietnam, by three different FDA District Offices, are the first refusals of Vietnamese shrimp for reasons related to banned antibiotics since May 2015. In total, the FDA has reported refusing 38 entry lines of Vietnamese shrimp for banned antibiotics this year, the second most since 2002 (48 entry lines of Vietnamese shrimp were refused for banned antibiotics in 2014).
The FDA’s findings are similar to those in other major seafood-importing markets.
For example, the Canadian Food Inspection Agency currently includes six Vietnamese companies on its Mandatory Inspection List (MIL) for antibiotics that were added in 2015:
- Nha Trang Seaproduct Company (Approval DL 17) was placed on the MIL on February 12, 2015 for both sulfonamides and fluoroquinolones;
- Global Blue Ocean Import Export Joint Stock Company (GLOCEAN BLUE) (EU Code DL 745) was placed on the MIL on March 4, 2015 for sulfonamides;
- Trang Thuy Private Enterprise (Plant Reg. No. HK337) was placed on the MIL on June 5, 2015 for amphenicols;
- Bien Dong Seafoods Co. Ltd. (EU# DL15) was placed on the MIL on September 18, 2015 for fluoroquinolones;
- Quoc Viet Seaproducts Processing Trading and Import-Export Co., Ltd. (DL 200) was placed on the MIL on November 23, 2015 for tetracyclines; and
- Vietnam Clean Seafood Corporation (VINA CLEANFOOD) (DL 707) was placed on the MIL on November 30, 2015 for amphenicols.
The Import Food Inspection Services (IFIS) of Japan’s Ministry of Health, Welfare and Labour has reported finding banned antibiotics in 23 shipments of shrimp from Vietnam in 2015, with rejections reported in every month of this year for a varied array of antibiotics:
- On January 21, 2015, IFIS published notice that a shipment of shrimp (frozen ebi fry) from Vietnam by New Wind Seafood Co. Ltd. was found to have traces of enrofloxacin;
- On January 30, 2015, IFIS published notice that a shipment of Stretched PTO Black Tiger Shrimp from Vietnam by Fine Foods Company was found to have traces of enrofloxacin;
- On February 10, 2015, IFIS published notice that a shipment of PTOE Black Tiger Shrimp from Vietnam by Seaprodex Minh Hai was found to have traces of enrofloxacin;
- On February 27, 2015, IFIS published notice that a shipment of Nobashi Ebi with STPP from Vietnam by Seaprimexco was found to have traces of enrofloxacin;
- On March 5, 2015, IFIS published notice that a shipment of Frozen Block Raw PDTO Vannamei Shrimp from Vietnam by Ngo Bros Seaproducts Import Export One Member Co., Ltd. was found to have traces of enrofloxacin;
- On March 13, 2015, IFIS published notice that a shipment of Vannamei Shrimp from Vietnam by Hoang Phuong Seafood Factory was found to have traces of furazolidone;
- On March 18, 2015, IFIS published notice that a shipment of Frozen Vanamei Shrimp Half Cut from Vietnam by Camau Seafood was found to have traces of oxytetracycline;
- On March 30, 2015, IFIS published notice that a shipment of Frozen Raw Sushi Ebi (Vanamei) from Vietnam by Vietnam Clean Seafood Corporation was found to have traces of sulfadiazine;
- On April 23, 2015, IFIS published notice that a shipment of Frozen Stretched PTO Black Tiger Shrimps from Vietnam by Fine Foods Company was found to have traces of enrofloxacin;
- On May 19, 2015, IFIS published notice that a shipment of Fresh Frozen PDTO Nobashi Vanamei Shrimp from Vietnam by Vietnam Clean Seafood Corporation was found to have traces of chloramphenicol;
- On May 19, 2015, IFIS published notice that a shipment of Frozen Vannamei Tempura from Vietnam by Ngo Bros Seaproducts Import Export One Member Co., Ltd. was found to have traces of enrofloxacin;
- On May 22, 2015, IFIS published notice that a shipment of Vannamei Deep Fry Shrimp from Vietnam by Saota Food Joint-Stock Co. was found to have traces of enrofloxacin;
- On June 2, 2015, IFIS published notice that a shipment of Frozen PDTO Nobashi Vannamei Shrimp from Vietnam by Minh Hai Joint-Stock Seafoods Processing Company was found to have traces of furazolidone;
- On June 2, 2015, IFIS published notice that a shipment of Kinchaku Shumai (with shrimp) from Vietnam by GN Foods Joint Stock Company was found to have traces of enrofloxacin;
- On June 22, 2015, IFIS published notice that a shipment of Vannamei Deep Fry Shrimp from Vietnam by Saota Food Joint-Stock Co. was found to have traces of enrofloxacin;
- On June 22, 2015, IFIS published notice that a shipment of Frozen Raw Sushi Ebi from Vietnam by Vietnam Clean Seafood Corporation was found to have traces of sulfadiazine;
- On July 24, 2015, IFIS published notice that a shipment of Frozen Raw Vannamei Shrimps from Vietnam by Seaprodex Minh Hai was found to have traces of furazolidone;
- On August 20, 2015, IFIS published notice that a shipment of Frozen Shrimp from Vietnam by Quoc Viet Seaproducts Processing Trading and Import-Export Co., Ltd. was found to have traces of furazolidone;
- On September 2, 2015, IFIS published notice that a shipment of Dried Baby Shrimp from Vietnam by Chin Tuy Pte. was found to have traces of chloramphenicol;
- On September 9, 2015, IFIS published notice that a shipment of Frozen Sushi Ebi Vannamei Shrimp from Vietnam by Ngo Bros Seaproducts Import Export One Member Company Limited was found to have traces of furazolidone;
- On October 13, 2015, IFIS published notice that a shipment of F Raw PD Vannamei Shrimp from Vietnam by Thuan Phuoc Seafoods and Trading Corporation was found to have traces of furazolidone;
- On October 28, 2015, IFIS published notice that a shipment of Frozen PD Black Tiger Shrimp from Vietnam by Cantho Import Export Fishery Limited Company (CAFISH VIETNAM) was found to have traces of enrofloxacin; and
- On November 19, 2015, IFIS published notice that a shipment of Frozen Shrimp Fry from Vietnam by Quoc Viet Seaproduct Processing Trading and Import-Export Co., Ltd. was found to have traces of enrofloxacin.
The European Union’s Rapid Alert System for Food and Feed (RASFF) includes six notifications issued in 2015 for Vietnamese shrimp contaminated with a variety of banned antibiotics:
- On January 14, 2015, a notification was issued from Germany regarding frozen shrimps (penaeus vannamei) from Vietnam found to have residues of chloramphenicol;
- On January 22, 2015, a notification was issued from Belgium regarding frozen sushi shrimps (penaeus vannamei) from Vietnam found to have residues of oxytetracycline;
- On January 30, 2015, a notification was issued from Denmark regarding frozen prawns (penaeus vannamei) from Vietnam found to have residues of tetracycline;
- On July 14, 2015, a notification was issued from Germany regarding frozen king prawns from Vietnam found to have residues of nitrofurans;
- On August 26, 2015, a notification was issued from Germany regarding frozen whiteleg shrimp (penaeus vannamei) from Vietnam found to have residues of oxytetracycline; and
- On September 1, 2015, a notification was issued from Sweden regarding frozen shrimps from Vietnam found to have residues of doxycycline.
Even Australia has reported issues with imports of Vietnamese shrimp this year. Australia’s Department of Agriculture and Water Resources publishes monthly “Failing Food” reports of results from its Imported Food Inspection Scheme, including a report that a shipment of marinated garlic prawns from Trang Khanh Seafood Co Ltd (DL 343) tested on May 22, 2015 was found to have ciprofloxacin.