In May, the U.S. Food and Drug Administration (FDA) reported that just 12 out of the 88 (13.6%) total seafood entry line refusals were of shrimp for reasons related to banned antibiotics.
Through the first five months of the year, the FDA has refused a total of 44 entry lines of shrimp for reasons related to veterinary drug residues, compared to 53 all of last year (and 55 in all of 2017). As summarized in the table below, the number of shrimp entry line refusals this year exceeds the annual refusals by the FDA reported between 2002 and 2006:
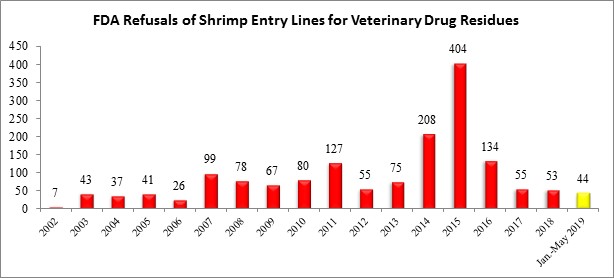
The twelve shrimp entry lines refused in May for veterinary drug residues were from two different exporters in Mexico and the United Arab Emirates:
- Dulceria Los Alamos S.A. de C.V. (Mexico), a company that was added to Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) on October 23, 2018 for its shipments of dried shrimp, had one entry line refused for shrimp contaminated with veterinary drug residues by the Division of Southwest Imports on May 28, 2019; and
- Freshly Frozen Foods (United Arab Emirates), a company that has been listed on Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) since June 27, 2018 for nitrofurans in its cooked shrimp shipments (with a separate listing for Freshly Frozen Foods Factory LLC), had eleven entry lines refused for shrimp contaminated with nitrofurans by the Division of Southwest Imports on May 7, 2019.
In addition, the FDA also reported that it had refused two entry lines of shrimp from Bangladesh and India for the presence of salmonella in April and another entry line of shrimp from Vietnam for salmonella in May.
