This morning, the U.S. Food and Drug Administration (FDA) published data reporting that there were 99 total seafood entry line refusals in May, of which 2 (2.0%) were of shrimp for reasons related to banned antibiotics. Of the remaining 97 seafood entry lines refused in March, 30 were of shrimp from Thailand found by the FDA to be either filthy or sanitary or contaminated with salmonella. The vast majority of these entry lines were from shrimp shipped by Sea Tech Intertrade Co. Ltd. (28), while entry lines of shrimp from the Thai companies N R Instant Produce Co. Ltd. and CK Frozen Fish & Food Co., Ltd. were also refused.
Through the first five months of this year, the FDA has now refused a total of 41 entry lines of antibiotic-contaminated shrimp.
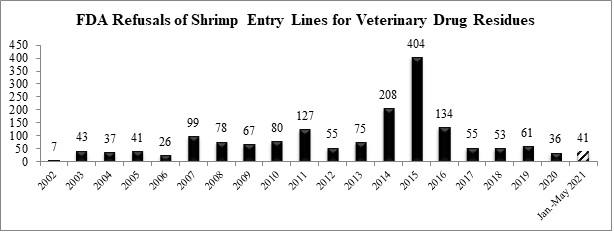
The two shrimp entry lines refused in May for reasons related to antibiotics were for shipments from Bangladesh and Vietnam:
- Rupsha Fish & Allied Industries Ltd. (Factory) (Bangladesh), a company that is currently listed twice on Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) as of February 5, 2021, had one entry line refused for shrimp contaminated with nitrofurans and veterinary drug residues by the Division of West Coast Imports on May 20, 2021; and
- Cadovimex Seafood Import-Export Processing (Vietnam), a company that is currently listed on Import Alert 16-124 (“Detention Without Physical Examination of Aquaculture Seafood Products Due to Unapproved Drugs”) as of June 9, 2017 for enrofloxacin, ciprofloxacin, and sulfadiazine for its shipments of tilapia, had one entry line refused for shrimp contaminated with nitrofurans and veterinary drug residues by the Division of West Coast Imports on May 18, 2021.
