Over the weekend, the U.S. Food and Drug Administration (FDA) published detailed data regarding 126 total seafood entry line refusals in August, of which 6 (4.8%) were of shrimp for reasons related to banned antibiotics. The FDA also released additional information regarding refusals of another 35 seafood entry lines in July, of which 5 were of shrimp for reasons related to banned antibiotics.
Including the additional refusals now reported for July, with four months left in the year, the FDA has refused a total of 61 entry lines of antibiotic-contaminated shrimp. As shown in the table below, this is the highest number of entry line refusals of shrimp for reasons related to veterinary drug residues since 2019 and the ultimate total in 2021 will be the highest since at least 2016.
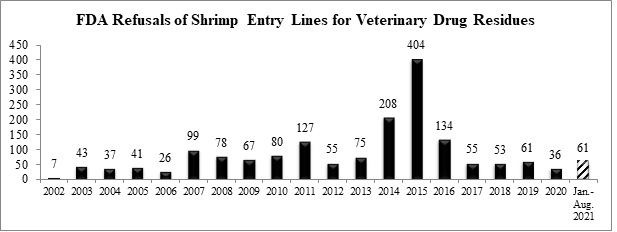
The five additional shrimp entry lines now reported as refused in July and the six shrimp entry lines refused in August for reasons related to antibiotics were for shipments from three different exporters in Bangladesh and Vietnam:
- Rupsha Fish & Allied Industries Ltd. (Factory) and Rupsha Fish & Allied Industries Limited (Bangladesh), a company that is currently listed twice on Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) as of February 5, 2021, had four entry lines refused, as Rupsha Fish & Allied Industries Ltd. (Factory), for shrimp contaminated with nitrofurans that were also described as filthy by the Division of West Coast Imports on July 30, 2021 and another five entry lines refused, as Rupsha Fish & Allied Industries Limited, for shrimp contaminated with veterinary drug residues and nitrofurans that was also described as being filthy and containing salmonella by the Division of Northeast Imports on August 6, 2021;
- Southern Foods Ltd. (Bangladesh), a company that is currently listed on Import Alert 16-124 (“Detention Without Physical Examination of Aquaculture Seafood Products Due to Unapproved Drugs”) as of February 18, 2021 for oxolinic acid in its shrimp exports and is listed twice on Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) as of August 30, 2016 and February 3, 2021, had one entry line refused for shrimp contaminated with veterinary drug residues and nitrofurans by the Division of West Coast Imports on August 10, 2021; and
- Can Tho Import Export Fishery Limited Company, aka CAFISH (Vietnam), a company that is currently listed on Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) as of March 4, 2021, had one entry line refused for shrimp contaminated with nitrofurans and veterinary drug residues by the Division of Southeast Imports on July 29, 2021.
