Over the holiday weekend, the U.S. Food and Drug Administration (FDA) published data reporting that there were 36 total seafood entry line refusals in June, of which one (2.8%) was of shrimp for reasons related to banned antibiotics.
The small number of total seafood entry lines refused in June (36) continues, for the second straight month, an unprecedented low in the agency’s history of oversight of imported seafood. Over the previous eighteen years (2002-2019), the FDA has refused an average of roughly 154 seafood entry in the month of June. The 36 seafood entry line refusals reported last month represents a drop of 76 percent below this historic average. The chart below sets out the total number of seafood entry lines refused by the FDA in the month of June for each of the last nineteen years.
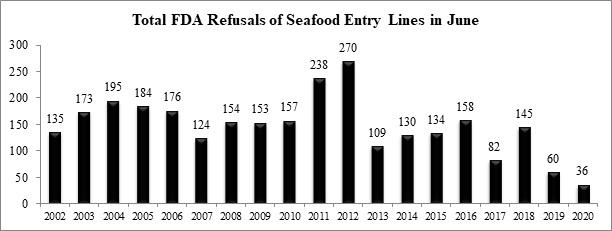
This remarkable decline in overall seafood entry line refusals implies a massive reduction in the FDA’s oversight activities regarding imported seafood at our ports of entry.
The FDA has reported a total of fifteen refusals of shrimp entry lines for reasons related to banned antibiotics through the first half of 2020.
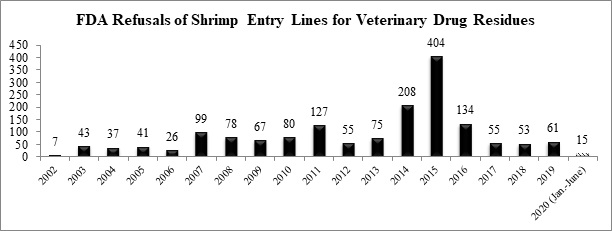
The shrimp entry line refused in June was for a shipment from India from a company that had also had refusals for shrimp contaminated with banned antibiotics in March and April:
- Ram’s Assorted Cold Storage Limited (India), a company that is currently listed on Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) as of October 10, 2019, had one entry line refused for shrimp contaminated with nitrofurans and veterinary drug residues by the Division of West Coast Imports on June 22, 2020.
In addition, the FDA reported refusing one additional entry line of shrimp shipped from India by Kader Exports Private Limited after detecting the presence of salmonella.
