On Sunday, the U.S. Food and Drug Administration (FDA) released details regarding imports refused entry into the United States for the month of May. In total, the agency reported refusing 179 seafood entry lines last month. Of these, 9 (5.0%) were of shrimp entry lines refused for reasons related to banned antibiotics.
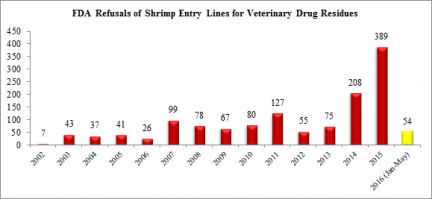
In the first five months of this year, the FDA has reported refusing almost as many shrimp entry lines for banned antibiotics (54) as in all of 2012 (55).
The nine entry line refusals in May involved two companies and were reported by two different regional offices of the FDA:
- Jagadeesh Marine Exports (India), a company not currently listed on Import Alert 16-129 for nitrofurans, but listed on Import Alert 16-124 for chloramphenicol on November 16, 2015, had eight entry lines refused for shrimp contaminated with veterinary drug residues in the San Francisco District; and
- Narong Seafood Co., Ltd. (Thailand), a company not currently listed on either Import Alert 16-124 or Import Alert 16-129 (there are no Thai companies on either of these Import Alerts), had one entry line refused for shrimp contaminated with veterinary drug residues and nitrofurans in the New England District. This was the first refusal reported by the FDA of a shrimp shipment from Thailand for banned antibiotics since September of 2005.
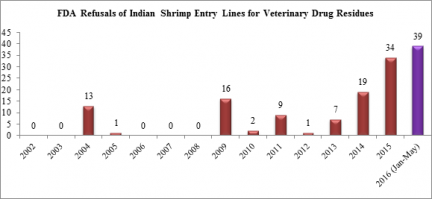
Nearly all of the shrimp entry line refusals in May were for shipments from India. Last year, a record total of 34 entry lines of shrimp from India were refused for reasons related to banned antibiotics. In 2016 thus far, the FDA has already reported 39 entry lines of shrimp from India refused for the same reasons.
After having only one entry line of shrimp refused for banned antibiotics in 2012, the number of entry line refusals for Indian shrimp contaminated with veterinary drug residues has increased significantly each of the last four years.