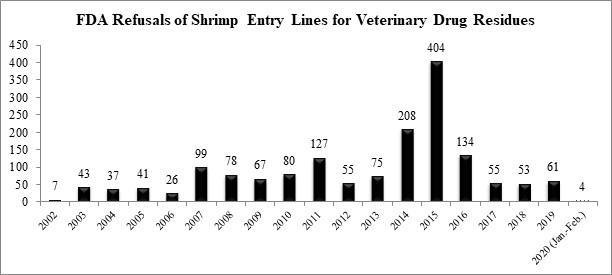
The U.S. Food and Drug Administration (FDA) announced yesterday that there were 97 total seafood entry line refusals in February, of which two were of shrimp for reasons related to banned antibiotics (2.1%).
The FDA has now reported a total of four refusals of shrimp entry lines for reasons related to banned antibiotics in 2020, well below the monthly average rate of ten refusals that the agency has reported over the last thirteen years.
The two shrimp entry lines refused in February were for the same two companies for whom refusals were reported by the FDA in January:
- Cochin Frozen Food Exports (India), a company that is currently listed on Import Alert 16-129 (“Detention Without Physical Examination of Seafood Products Due to Nitrofurans”) as of March 4, 2020, had one entry line refused for shrimp contaminated with veterinary drug residues by the Division of West Coast Imports on February 14, 2020; and
- Dalian Zhuohong Marine Product Co., Ltd. (China), a company that has not been green-listed on Import Alert 16-131 (“Detention Without Physical Examination of Aquacultured, Shrimp, Dace, and Eel from China – Presence of New Animal Drugs and/or Unsafe Food Additives”), had one entry line refused for shrimp contaminated with veterinary drug residues and an unsafe additive by the Division of Northeast Imports on February 3, 2020.
In addition to those two entry lines, the FDA also refused another six entry lines of shrimp because of the presence of salmonella: four from Bangladesh (three from Modern Seafood Industries Limited and one from Bright Seafood Limited), all by the Division of West Coast Imports; one from India (Choice Canning Company), by the Division of Northeast Imports; and one from Thailand (CK Frozen Fish & Food Co., Ltd.), also by the Division of Northeast Imports.
